How Quality Control, Quality Assurance And A QMS Fit Together

It’s a strategic priority. Whether you’re building cars, delivering healthcare, developing software, or manufacturing medical devices, quality determines satisfaction, compliance, profitability, and reputation. Every organisation that wants consistent, reliable outputs must understand three interlinked concepts:
-
Quality Control (QC)
-
Quality Assurance (QA)
-
Quality Management System (QMS)
At first glance, the terms can seem interchangeable. Yet they represent distinct functions within a broader quality ecosystem that, when integrated effectively, empower organisations to prevent issues, detect defects, learn from feedback, and constantly improve. This blog explains how these pieces fit together, how they differ, and why each is necessary for a mature, high-performance system.
1. What Is Quality? A Baseline Definition
Before diving into QC, QA, and QMS, let’s define quality itself.
Quality is typically defined as the degree to which a product or service meets defined requirements and customer expectations. In practical terms, quality means:
-
Delivering what was promised
-
On time
-
Without defects
-
And in a way that satisfies customers and regulations
The pursuit of quality is not a one-time activity — it’s a continuous commitment that spans planning, execution, monitoring, and improvement.
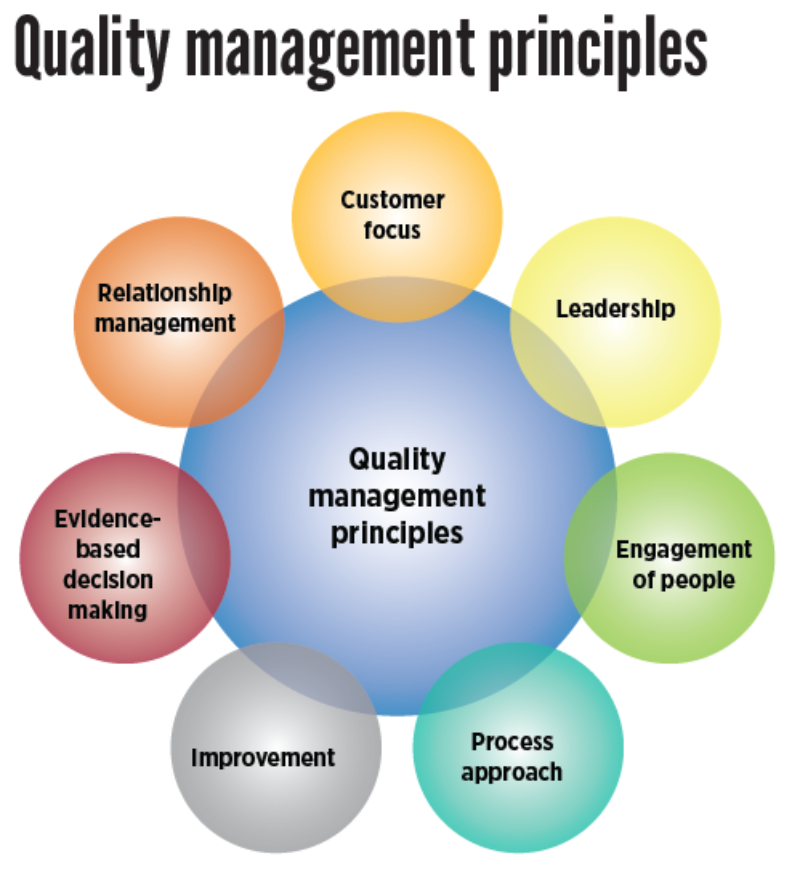
2. Quality Management System (QMS): The Big Picture
2.1 Definition and Core Purpose
A Quality Management System (QMS) is the overarching framework that organisations use to manage and control all activities affecting quality. At its core, a QMS is:
A structured system of policies, processes, procedures, resources, and responsibilities designed to ensure that products and services consistently meet customer requirements and regulatory standards while enabling continuous improvement.
In other words, the QMS is not just about checking outputs it’s about embedding quality into everything an organisation does.
2.2 What a QMS Includes
A robust QMS typically contains:
-
Quality policy and objectives: High-level commitments set by leadership
-
Documented procedures: Standard operating procedures (SOPs), work instructions
-
Process controls and monitoring: How activities are tracked and measured
-
Performance metrics: Key performance indicators (KPIs) related to quality
-
Corrective and Preventive Actions (CAPA): Systems to fix current issues and prevent future ones
-
Audits and reviews: Internal and external assessments
-
Training and competence: Ensuring staff know what to do
-
Risk management: Identifying, evaluating, and mitigating risks to quality
In regulated environments such as medical devices, aerospace, or pharmaceuticals — QMS requirements may be mandated by standards like ISO 9001, ISO 13485, EU regulations, or FDA 21 CFR Part 820.
2.3 QMS Is the “Umbrella” System
The QMS is the umbrella under which QA and QC sit:
-
It defines how quality activities occur
-
It ensures that those activities are systematic, documented, and measurable
-
It ties quality work back to strategic objectives and continuous improvement
In many organisations, the QMS is supported by software platforms often called electronic QMS (eQMS) that automate documentation, track corrective actions, manage audits, and integrate QA/QC data.
3. Quality Assurance (QA): Preventing Problems Before They Happen
3.1 What Is Quality Assurance?
Quality Assurance is proactive: it ensures that processes are designed and implemented in a way that prevents quality problems from occurring. Unlike inspection, which reacts to results, QA establishes the conditions for quality at the outset.
Put simply:
QA defines how quality will be achieved through planned, systematic activities that make defects unlikely in the first place.
3.2 The Role of QA Within a QMS
Within the QMS framework:
-
QA creates the standards, procedures, and process controls
-
It ensures that people know what to do and how to do it
-
It verifies compliance through audits, training, and process validation
-
It embeds risk management and process design thinking into daily work
In mature systems, QA is not just a department it’s a culture that emphasises prevention, learning, and structured improvement.
3.3 QA Is System-Focused and Preventive
Quality Assurance:
-
Is process-oriented
-
Happens before and during production
-
Aims to prevent defects
-
Covers organisational systems, procedures, training, and controls
In contrast, QA is less about inspecting outputs and more about asking:
-
Are our processes well designed?
-
Are workers trained and equipped?
-
Do we have controls that prevent errors at source?
4. Quality Control (QC): Detecting and Correcting Problems
4.1 What Is Quality Control?
Quality Control is reactive: it involves inspecting products, services, or outcomes to check whether they meet defined quality standards. QC detects defects that may have slipped through even well-designed processes.
In simple terms:
QC checks what has been produced and ensures it conforms to requirements.
4.2 How QC Fits Within a QMS
Within a QMS, Quality Control involves:
-
Inspection and testing of products or deliverables
-
Sampling plans and acceptance criteria
-
Measurement and recording of results
-
Reporting non-conformances
-
Triggering CAPA when problems are detected
Quality Control ensures that outputs whether products or services are safe, functional, and compliant before they reach the customer.
4.3 QC Is Product-Focused and Reactive
Key characteristics of QC:
-
Product-oriented: It looks at outputs
-
Reactive: It identifies issues that have already occurred
-
Specific: Often involves inspection, testing, sampling, and statistical checks
While QA seeks to prevent defects, QC seeks to find and fix them. In many systems, QC data becomes input for QA process improvements and QMS adjustments.
5. How QC, QA and QMS Fit Together
Now that we’ve defined each term, let’s see how they interact in a mature quality environment.
5.1 A Hierarchy of Quality Activities
One way to visualise the relationship is as a layered approach:
-
QMS (system level): Defines the entire quality ecosystem policies, processes, metrics, and improvement cycles.
-
QA (process level): Uses the QMS framework to design processes that deliver quality.
-
QC (output level): Uses inspection and testing to confirm that products/services conform to standards.
In this sense:
-
QC is part of QA
-
QA is part of the QMS
-
But in practice, all three interact dynamically
As one quality expert notes:
QC provides on-the-ground tests to ensure QA plans and processes are working properly, while the QMS houses and coordinates it all
5.2 Proactive + Reactive + System Thinking
These three functions create a complete quality picture:
-
QMS ensures that the right things are done in the right way
-
QA ensures processes are designed to prevent defects
-
QC ensures outputs actually meet standards
Without QA, QC would just react to problems.
Without QC, QA couldn’t verify whether processes deliver promised quality.
Without a QMS, both would be uncoordinated, inconsistent, and less effective.
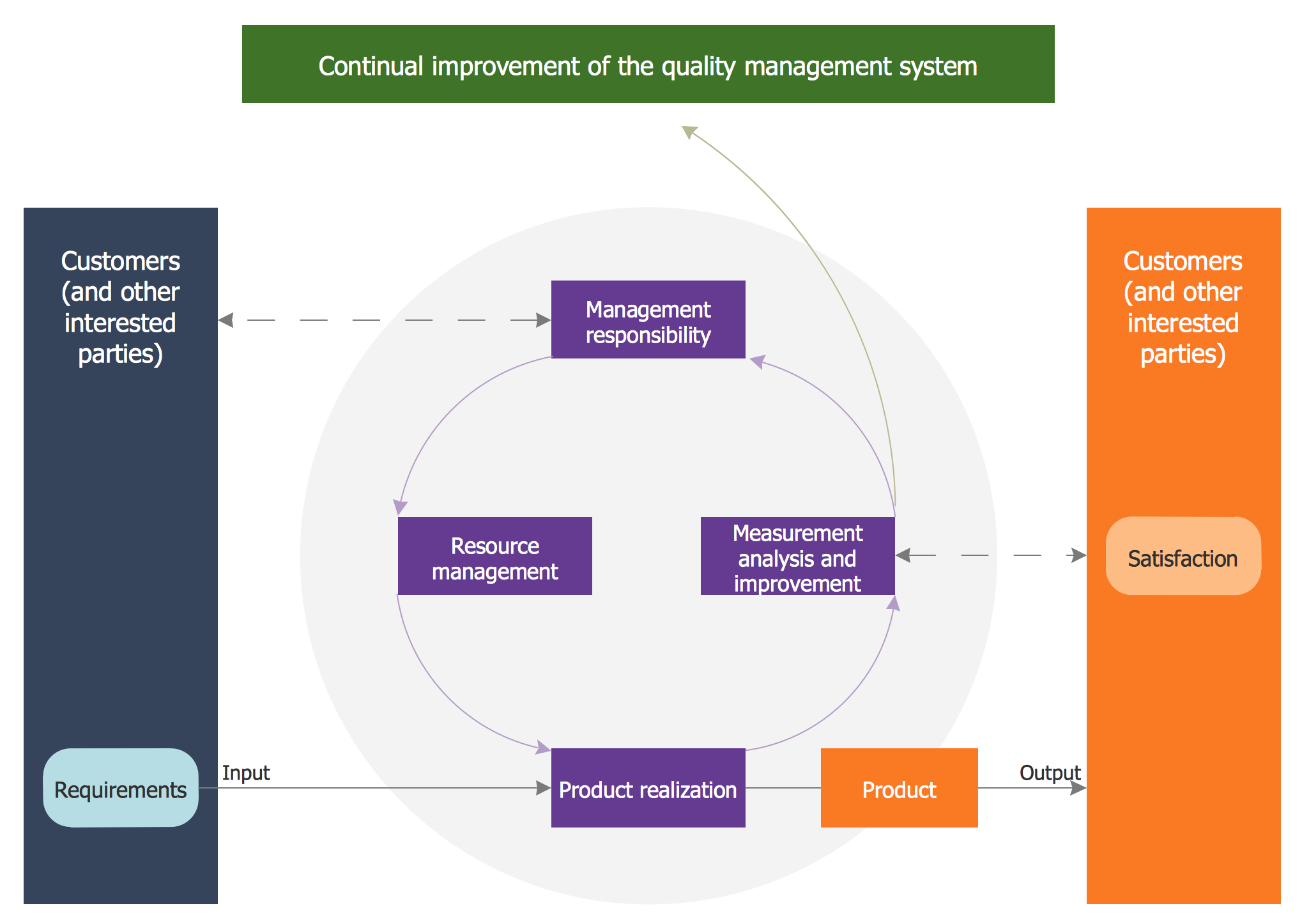
5.3 Feedback Loops and Continuous Improvement
Integration is not static it depends on feedback loops:
-
QC findings trigger CAPA and feed process improvements
-
QA metrics inform strategic QMS reviews
-
The QMS adjusts standards based on customer feedback, audit results, and defect trends
This closed-loop system is central to continuous improvement especially in ISO 9001 and other standards that emphasise Plan–Do–Check–Act (PDCA) cycles.
6. Common Misconceptions
Understanding how these terms relate helps avoid common pitfalls:
6.1 “QA and QC Are the Same”
They are related but distinct:
-
QA focuses on process
-
QC focuses on product
-
Both fit under a QMS but serve different purposes
This distinction is critical for role clarity and organisational effectiveness.
6.2 “More Inspection Means Better Quality”
Relying solely on QC is inefficient. Inspection finds defects it does not prevent them. Effective QA and well-designed processes reduce the need for costly rework.
6.3 “A QMS Is Just Documentation”
While documentation is important, a QMS is a living system of practices that drive real performance, not just paperwork.
7. Practical Examples
To illustrate the concepts:
7.1 Manufacturing
-
QMS: ISO 9001 certified system with defined SOPs, training plans, audit schedules
-
QA: Process validation, equipment calibration schedules, worker training programs
-
QC: Final product inspection, dimension measurements, batch testing
A QC failure triggers corrective action: if too many defective units appear, QA reviews the process to prevent recurrence.
8. Benefits of Integrating QC, QA and QMS
When fully integrated, organisations enjoy:
| Benefit | How It Happens |
|---|---|
| Better quality products | QA designs robust processes and QC verifies outcomes |
| Higher customer satisfaction | Consistency and reliability build trust |
| Regulatory compliance | QMS ensures documentation and auditable standards |
| Lower costs | Less rework, fewer defects, fewer warranty claims |
| Continuous improvement | Feedback loops drive ongoing enhancements |
9. Best Practices for Integration
To maximise impact:
9.1 Clarify Roles & Responsibilities
Ensure that all staff understand how QC, QA, and QMS interact.
9.2 Establish Clear Metrics
Track process performance, defect rates, audit findings, and improvement initiatives.
9.3 Use a Unified QMS Platform
Adopt a centralised system (often digital) to manage QA and QC data, documentation, and reporting.
9.4 Embrace Continuous Improvement
Use CAPA and PDCA cycles to turn data into action.
9.5 Align Leadership and Culture
Quality must be supported from the top down and embraced across functions.
10.
Quality does not happen by accident it’s the result of deliberate systems, processes, checks, and culture. A Quality Management System (QMS) provides the strategic framework. Quality Assurance (QA) embeds preventive, process-focused activities. Quality Control (QC) verifies that outputs meet standards and informs improvement.
Together, they form a powerful, integrated approach that turns quality into a competitive advantage rather than a costly burden. Whether you’re aiming for ISO certification, regulatory compliance, or customer delight, understanding how these elements fit together is essential for organisational excellence and long-term success.